The Rosalia longicorn Rosalia alpina (LINNAEUS, 1758) |
Text © Klaas Reißmann, 2010
|
|
|
1. Introduction |
|
The Rosalia longicorn is probably one of the best known beetles of the European Fauna, not only due to its beautiful black and blue coloration and the
striking black tufts of hair on the central segments of the antennae. With a body length of up to 40 mm it is also among the largest beetles of our fauna.
Due to its dramatic decline in the recent decades, Rosalia alpina has moved into the focus of public attention. The intensive use of the beech forests
for the production of firewood and furniture wood, but also a mistaken forest management with its rigorous removal of any dead wood, have deprived the Rosalia
longicorn of its livelihood in large parts of Europe and brought it to the brink of extinction.

Female of Rosalia alpina. Note the shorter and thicker antennae with tufts of hair up to the 7th segment and the plain mandibles
The marvelous coloration of the Rosalia longicorn reveals itself only under a magnifying glass. In principle, the beetle is plain black. At the basis of the
elytra a number of shiny black grains are noticeable to the naked eye. The largest part of the body is covered with a dense tomentum, consisting of
very fine, light blue, blue gray and dark blue hairs. The black spots on the pronotum and the elytra are covered with tomentum as well, consisting of fine and dense
plain black hairs which give the spots their velvety appearance.

Male of Rosalia alpina. Note the longer and thinner antennae with tufts of hair up to the 5th segment and the toothed mandibles
The nominotypical taxon is blue colored with three black spots on each elytron and one black spot in the middle of the anterior border of the pronotum.
The size of these spots varies considerably. They can be extended or reduced, partly or totally merged or may be completely absent.
 Johann Jakob SCHEUCHZER (1672-1733) and Carl von LINNÉ (1707-1778)
Johann Jakob SCHEUCHZER (1672-1733) and Carl von LINNÉ (1707-1778)
The enormous variability also led to the description and designation of more than 100 aberrations. Among the most conspicuous, but also rarest aberrations are extreme forms
like Rosalia alpina a. unicolor, which is of plain blue color, and Rosalia alpina a. croissandeaui, which is almost totally black. Moreover,
there are forms with pale rose coloration instead of blue.
Carl von LINNÉ, the founder of the binary nomenclature, described Rosalia alpina in 1758 for the first time from a specimen
collected by Johann Jakob SCHEUCHZER on July 12th, 1703 in the Tamina valley between Valens and Vättis (Sarganserland, Switzerland),
which belongs today to the modern canton of St. Gallen. The species name "alpina" refers to this location. It is derived from Latin and means
"living on the alps", although this is misleading to a certain extent, as the distribution of Rosalia alpina is by far not restricted to the Alps.
|
2. Distribution |
|
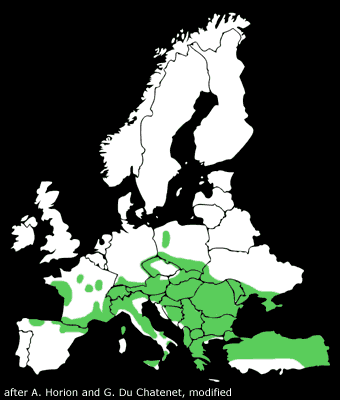 The distribution of the Rosalia longicorn stretches over Central and Southern Europe. The western border of the range is marked by the Pyrenees. The
southwesternmost records originate from the Spanish provinces of Asturias, Cantabria, the Basque Country, Navarre, Aragon and Catalonia.
VILLERS (1946) does not mention the species for North Africa. DEMELT (1957) cites Rosalia alpina for North Africa from the Atlas Mountains,
but without mentioning a source and does also not refer to VILLERS' Fauna of North Africa - presumably out of ignorance. SAMA (2002),
a distinguished expert of the North African longhorn beetle fauna, judges the report as erroneous (BENSE, written notice). Therefore Rosalia alpina
should be deleted from the North African species list, although HORION (1974) does mention the species for North Africa.
Rosalia alpina is known to occur to the southern tip of Italy, also on the islands Sicily and Corsica, but not - contrary to other peoples' opinion - on Sardinia.
In Southeast Europe, the range stretches from Greece and Bulgaria to Southeastern Turkey, where the species is represented by the subspecies Rosalia alpina syriaca
PIC, 1894. According to SAMA (2002) the occurrences in Southeastern Turkey are probably only relics and the species might be already extinct there.
Various authors also cite Jordan, Israel, Palestine and Syria. According to the recent Fauna of Israel by SAMA (2002) and SAMA et al. (2010)
this seems to be incorrect (BENSE, written notice) and is at least questionable. In the northeast of Turkey the border of the distribution range is marked by
the Caucasus, although the beetle seems to occur around the whole Black Sea. The northern limit of the distribution runs through Northern France, where the occurrence
is only insular and isolated, through Southern Germany and Czechia to Poland. Poland also marks the northeastern limit of distribution.
In Eastern Europe, the distribution stretches over Slovakia, Hungary and the Ukraine to Russia. LAGUNOV and NOVOZHENOV reported the species in
1996 for the vicinity of Yekaterinburg (formerly Sverdlovsk) from the Ilmen natural reserve (Western Siberia, central Ural Mountains).
SHAPOVALOV collected a specimen on July 13th, 2008 in the Orenburg region in the Southern Ural Mountains (BENSE, written notice).
Hence, the Central and South Ural Mountains constitute the eastern limit of the distribution of Rosalia alpina.
According to Horion, at the beginning of the 20th century there was a settlement in Denmark on the island Sjælland and in South Sweden for a brief period of time.
The last record was made in 1926 for Västergötland.
The distribution of the Rosalia longicorn stretches over Central and Southern Europe. The western border of the range is marked by the Pyrenees. The
southwesternmost records originate from the Spanish provinces of Asturias, Cantabria, the Basque Country, Navarre, Aragon and Catalonia.
VILLERS (1946) does not mention the species for North Africa. DEMELT (1957) cites Rosalia alpina for North Africa from the Atlas Mountains,
but without mentioning a source and does also not refer to VILLERS' Fauna of North Africa - presumably out of ignorance. SAMA (2002),
a distinguished expert of the North African longhorn beetle fauna, judges the report as erroneous (BENSE, written notice). Therefore Rosalia alpina
should be deleted from the North African species list, although HORION (1974) does mention the species for North Africa.
Rosalia alpina is known to occur to the southern tip of Italy, also on the islands Sicily and Corsica, but not - contrary to other peoples' opinion - on Sardinia.
In Southeast Europe, the range stretches from Greece and Bulgaria to Southeastern Turkey, where the species is represented by the subspecies Rosalia alpina syriaca
PIC, 1894. According to SAMA (2002) the occurrences in Southeastern Turkey are probably only relics and the species might be already extinct there.
Various authors also cite Jordan, Israel, Palestine and Syria. According to the recent Fauna of Israel by SAMA (2002) and SAMA et al. (2010)
this seems to be incorrect (BENSE, written notice) and is at least questionable. In the northeast of Turkey the border of the distribution range is marked by
the Caucasus, although the beetle seems to occur around the whole Black Sea. The northern limit of the distribution runs through Northern France, where the occurrence
is only insular and isolated, through Southern Germany and Czechia to Poland. Poland also marks the northeastern limit of distribution.
In Eastern Europe, the distribution stretches over Slovakia, Hungary and the Ukraine to Russia. LAGUNOV and NOVOZHENOV reported the species in
1996 for the vicinity of Yekaterinburg (formerly Sverdlovsk) from the Ilmen natural reserve (Western Siberia, central Ural Mountains).
SHAPOVALOV collected a specimen on July 13th, 2008 in the Orenburg region in the Southern Ural Mountains (BENSE, written notice).
Hence, the Central and South Ural Mountains constitute the eastern limit of the distribution of Rosalia alpina.
According to Horion, at the beginning of the 20th century there was a settlement in Denmark on the island Sjælland and in South Sweden for a brief period of time.
The last record was made in 1926 for Västergötland.
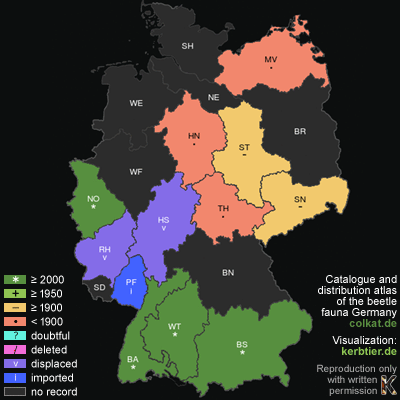
For reports of occurrences in Germany we need to take a very nuanced view. What is certain is that the old records from Rhineland Palatinate, Hesse and
the Rhine Province are based on displaced animals. Historic records from Lower Saxony (namely from the region of Hannover), Mecklenburg-Western Pomerania,
Saxony, Saxony-Anhalt and Thuringia are attributed by various authors to autochthonous populations. Other authors however, ascribe these records to displaced
animals with subsequent settlement for a brief period of time. In comparison with the scientifically proven occurrences in Baden-Württemberg and Bavaria,
the latter opinion seems plausible, given to large distance of the isolated occurrences in Lower Saxony and Mecklenburg-Western Pomerania from the autochthonous
locations. Also these locations do not match the requirements Rosalia alpina has for its habitats.
Arguably, occurrences in Saxony, Saxony-Anhalt and Thuringia would bridge the gap to the northeastern areas in Poland and seem to be
explicable by a potential connect to the occurrences in the Czech Republic. However, also these locations do not match the requirements of Rosalia alpina regarding
its habitats in Central Europe. HORION (1974) explains the displacement by the transport of logs over the large rivers like Oder or the Elbe.
In this way a short-term settlement could be logically explained. However, the occurrences in Lower Saxony (Hannover) and Mecklenburg-Western Pomerania
could be regarded as autochthonous only if the range of the Rosalia longicorn was covering the Northern areas in former times and the species slowly retreated
due to increasingly unfavorable climate conditions. This theory is not plausible, as the locations in question do not (and never did) exhibit favorable warm climate
conditions. The only surely established records of autochthonous occurrences in Germany remain those from Baden-Württemberg, in major parts of the
Swabian Alb and from Bavaria in parts of the Alpine region.
|
3. Lifestyle |
|
In Central Europe Rosalia alpina prefers sparse, primeval beech forests on south- or west-facing slopes, from the montane to the subalpine region
up to 1500 m ASL, most preferably between 600 and 1000 m ASL. The imagines appear from end of June until early September, with a maximum of activity between
mid of July and mid of August. In Central Europe the development takes place in the wood of dead or dying, sun-exposed beeches (Fagus sylvatica),
very rarely in sycamore (Acer pseudoplatanus). BENSE (1995) mentions for Southern Europe apart from beech (Fagus) and sycamore (Acer)
also elm (Ulmus), willow (Salix), chestnut (Castanea), ash (Fraxinus), walnut (Juglans), linden (Tilia), oak (Quercus),
alder (Alnus) and hawthorn (Crataegus). Sun-exposed dead or decaying trees, dead branches, or spots of damage in otherwise
healthy trees are suitable for development. In the tree top, branches of approx. 10 cm are accepted, otherwise the beetle prefers larger structures of at least 20 cm diameter.
The development cycle can be successfully concluded in dry and decaying wood. Such conditions are normally found in standing deadwood, which can be populated
over a long period of time, up to 10 years as substrate. Lying deadwood is only suitable, if sun-exposed and reasonably dry. Otherwise, the progressive
infestation with mould fungi prevents a successful development, similar to deep-cut tree stumps.

 The male occupies a small territory on suitable breeding substrate and defends it against competing males in ritualized fights. After mating, the female deposits
its eggs singly in bark cracks and dry splittings in wood, preferably in sun-exposed areas. The total number of eggs laid by the female is unknown. The larvae
feed in the sapwood, mainly at the edge between sapwood and heartwood. They usually avoid the nutrient-poor heartwood. The development cycle takes three to four years,
but can be completed in only two years under optimal conditions. During spring and early summer the larva produces a burrow for hatching, which is subsequently
sealed with wood shavings. The arch-shaped puparium is build close under the surface and is used for pupation only. After hatching, the adult beetles reside on the breeding substrate.
They have a lifespan of three to six weeks. The beetles feed on tree sap and can also be observed feeding on foliage. On rare occasions, the beetles have been observed
on blossoms, e.g. on wild carrot (Daucus carota). Like other longhorn beetles, the Rosalia longicorn is an average flyer. On warm days the beetles swarm during the hot hours
around noon. The beetles fly around their breeding trees or search for new breeding trees or nearby logs. The spatial spreading is most probably low, in most cases the covered
distance is below 1000 m.
The male occupies a small territory on suitable breeding substrate and defends it against competing males in ritualized fights. After mating, the female deposits
its eggs singly in bark cracks and dry splittings in wood, preferably in sun-exposed areas. The total number of eggs laid by the female is unknown. The larvae
feed in the sapwood, mainly at the edge between sapwood and heartwood. They usually avoid the nutrient-poor heartwood. The development cycle takes three to four years,
but can be completed in only two years under optimal conditions. During spring and early summer the larva produces a burrow for hatching, which is subsequently
sealed with wood shavings. The arch-shaped puparium is build close under the surface and is used for pupation only. After hatching, the adult beetles reside on the breeding substrate.
They have a lifespan of three to six weeks. The beetles feed on tree sap and can also be observed feeding on foliage. On rare occasions, the beetles have been observed
on blossoms, e.g. on wild carrot (Daucus carota). Like other longhorn beetles, the Rosalia longicorn is an average flyer. On warm days the beetles swarm during the hot hours
around noon. The beetles fly around their breeding trees or search for new breeding trees or nearby logs. The spatial spreading is most probably low, in most cases the covered
distance is below 1000 m.
|
4. Incidence |
|
In former times the Rosalia longicorn was so common in Central and Southern Europe, that the beetle was displaced with logs every once in a while, e.g. in 1926
to Västergötland in Sweden. In Czechia the beetle was widespread in beech forests. Today there's one population left at the Rollberg in Northern Bohemia and six
populations in Central and Eastern Czechia. The metapopulation at the Rollberg seems to consist of three subpopulations, of which one is healthy, the second
vulnerable and the third is regarded as threatened with extinction. Altogether Rosalia alpina is critically endangered in Czechia and is consequently strictly
protected by law. In Slovakia the situation is different. Numerous primeval beech forests are present countrywide and more than 50 populations of Rosalia alpina are known
(SLÁMA 1998). Nevertheless, also in Slovakia the species enjoys the highest protection category.
In Austria, the loss of habitats is massive. Judging from GEPP's map (2002), the records in Austria declined by more than 80% since 1980.
The decline in records can be also attributed to the fact that many coleopterists refrain from publishing their findings due to the legal protection of the species
in the late 1970s and the legal consequences for catching it. Moreover, the massive loss of suitable habitats and hence occurrences in Austria cannot be overlooked.
In the Red List of Endangered species of Carinthia, STEINER (1999) cites Rosalia alpina as vulnerable ("Vorwarnliste"),
contrary to the statement of GEPP (2002), which indicates a threat more severe than would be justified for the status "vulnerable".
The circumstances in Carinthia have not changed much. Neither did they improve, nor did they worsen, since the species can be observed regularly in the known habitats.
However, the traditional harvest and storage of beech logs and the building of woodstacks at sun-exposed spots may lead to a high loss of potential individuals
and thus may further weaken the population.
 In Germany, the Rosalia longicorn already disappeared in the 19th century after a short settlement from the Hannover region and Mecklenburg-Western Pomerania.
The last records from Saxony, Saxony-Anhalt and Thuringia were made before 1950. Whether these records are based on autochthonous occurrences is disputed and at least in the case
of the Hannover region and Mecklenburg-Western Pomerania doubtful (see Chapter 2 "Distribution"). From the Swabian Alb the species was recorded from the region between
Immendingen and Sigmaringen, a settlement area, from which v. d. TRAPPEN already in 1933 reported a critical threat for the species due to cutting of overaged beeches.
Today, Rosalia alpina still occurs in the above-mentioned area, but suffered severe declines since v. d. TRAPPEN until 1975.
Since then, the species at least did not disappear from the known locations in the Swabian Alb, the populations are stagnant. Since about 1993, the populations on the Swabian
Alb seem to recover. However, any spatial spreading is not discernible (BENSE et al. 2003). Records from Baden originate exclusively from the time before 1950,
although Rosalia alpina is also currently recorded from the historically known areas. The latter are assigned to Württemberg today. In Bavaria the situation looked extremely
bad until the 1980s. Until 1980, there was a loss of habitats of close to 30% observed, and another 40% during the 1980s, relative to the populated areas known in 1980.
Only around 1990 the populations became stagnant. Since around 10 years, the populations recover noticeably (BENSE et al. 2003). In the Red List of Germany
the Rosalia longicorn is classified as endangered (RL 2) (BINOT 1998).
In Germany, the Rosalia longicorn already disappeared in the 19th century after a short settlement from the Hannover region and Mecklenburg-Western Pomerania.
The last records from Saxony, Saxony-Anhalt and Thuringia were made before 1950. Whether these records are based on autochthonous occurrences is disputed and at least in the case
of the Hannover region and Mecklenburg-Western Pomerania doubtful (see Chapter 2 "Distribution"). From the Swabian Alb the species was recorded from the region between
Immendingen and Sigmaringen, a settlement area, from which v. d. TRAPPEN already in 1933 reported a critical threat for the species due to cutting of overaged beeches.
Today, Rosalia alpina still occurs in the above-mentioned area, but suffered severe declines since v. d. TRAPPEN until 1975.
Since then, the species at least did not disappear from the known locations in the Swabian Alb, the populations are stagnant. Since about 1993, the populations on the Swabian
Alb seem to recover. However, any spatial spreading is not discernible (BENSE et al. 2003). Records from Baden originate exclusively from the time before 1950,
although Rosalia alpina is also currently recorded from the historically known areas. The latter are assigned to Württemberg today. In Bavaria the situation looked extremely
bad until the 1980s. Until 1980, there was a loss of habitats of close to 30% observed, and another 40% during the 1980s, relative to the populated areas known in 1980.
Only around 1990 the populations became stagnant. Since around 10 years, the populations recover noticeably (BENSE et al. 2003). In the Red List of Germany
the Rosalia longicorn is classified as endangered (RL 2) (BINOT 1998).
In large parts of Europe, Rosalia alpina was in retreat. The reasons are clearly evident. On the one hand, a mistaken forest management with its rigorous removal of ailing and
dead trees not only to meet road-safety obligations but also a tendency to "clean up" forests has contributed to this decline. Cleared beech forests have been recultivated with fast-growing
pine monocultures or other trees. On the other hand, one of the major problems today remains the production of firewood from beech. The logging itself is not the real issue, but the temporary storage
of the logs in woodstacks, especially in locations close to the forest on sun-exposed spots. These woodstacks are highly attractive to the beetles as they contain both large amounts
and large-dimensioned wood, which is used by the females for egg deposition. The effectiveness of such woodstacks as traps should not be underestimated. Larvae hatching from
eggs deposited on such stacks complete their development only rarely. Most of them end long before in sawmills and furnaces.
|
5. Protection |
|
 The Rosalia longicorn is listed on Annex II of EU Council regulation 92/43/EWG and is classified as a priority species. Hence the European Community carries a particular
responsibility for the protection and preservation of the species, also as far as its habitats are concerned. Furthermore, the species is protected by Annex II of the Berne Convention of
1979 and enjoys protection Europe-wide. That means that it is strictly forbidden to capture, kill, possess or trade beetles or parts of the beetles, as well as their development
stages or parts thereof. Also existing populations and their habitats enjoy strict protection. According to the Federal Regulation for the Protection of Species Rosalia alpina
is protected as well, as most representatives of the longhorn beetles (family Cerambycidae) are protected, except a few varmints.
The Rosalia longicorn is listed on Annex II of EU Council regulation 92/43/EWG and is classified as a priority species. Hence the European Community carries a particular
responsibility for the protection and preservation of the species, also as far as its habitats are concerned. Furthermore, the species is protected by Annex II of the Berne Convention of
1979 and enjoys protection Europe-wide. That means that it is strictly forbidden to capture, kill, possess or trade beetles or parts of the beetles, as well as their development
stages or parts thereof. Also existing populations and their habitats enjoy strict protection. According to the Federal Regulation for the Protection of Species Rosalia alpina
is protected as well, as most representatives of the longhorn beetles (family Cerambycidae) are protected, except a few varmints.
However, in most cases the sylvicultural exploitation of the protected areas is allowed. In this way, the protection becomes meaningless, as every year large parts of the existing
population are displaced via woodstacks from the conservation area and their habitat and the larvae cannot complete their development cycle successfully.
Investigations by the swiss confederate research institute WSL have shown that males and females reside preferentially on long, sun-exposed logs with at least 25 cm diameter.
As a consequence the researches propose to allocate parts of beech logs of at least 25 cm diameter near woodstacks on sun-exposed spots. These logs are capable to lure at least
a larger part of the females away from the woodstacks. In this case egg deposition takes place on these log pieces. After two to three years of exposition the log pieces
should be deposited at sunny edges of forests, where the beetles hatch and are more or less kept away from woodstacks.
Based on these results, the Swiss association Pro Natura announced a both simple and ingenious, however probably quite expensive concept.
The association offers a compensation of 100 Swiss francs to all persons that put up beech log pieces in areas, where Rosalia alpina is known to occur
(DUELLI & WERMELINGER 2005). Critics however claim that this concept might overlay the existing structural problems
and may rather do more harm than good with regard to habitat protection. To really protect the endangered species, it seems necessary to start the inconvenient process of rethinking
and to quit the traditional production of (fire-) wood in habitats of the Rosalia longicorn. Furthermore, a well-blended age structure of the trees within the habitat, i.e.
a mosaic of tree stocks of different age would be desirable. As might be expected such a rethinking would be difficult to achieve. Therefore, tree cutting in protected areas should be tied to
legal restrictions: Existing dead wood must remain in the conservation area, woodstacks should be placed in shaded spots or at a sufficient distance from the habitat.
Further regulations could include the obligation to keep tree stumps of at least 1.5 m height when clearing. Also the planting of solitary trees in meadows could help. In Central
Europe Common beech (Fagus silvatica) and possibly sycamore (Acer pseudoplatanus) would be most suitable.
|
Thanks |
|
The author wishes to express his thanks to Mr. Ulrich BENSE for his detailed information on the distribution of the Rosalia longicorn. Without his support and advice
the chapter on the distribution would have been by far less precise. Furthermore we thank Mr. Martin REJZEK for his data regarding occurrences in Czechia and Slowakia,
Mr. Manfred EGGER for additional detailed information and Ms. Irina WÜRTELE for her critical review of the draft.
|
Sources |
Literature:
- BENSE, U.: "Bockkäfer", illustrierter Schlüssel zu den Cerambyciden und Vesperiden Europas, Margraf Verlag
- BENSE, U. et al. (2003): 4.10 Rosalia alpina (LINNAEUS, 1758). In: PETERSEN, B., ELLWANGER, G.,
BIEWALD, G., HAUKE, U., LUDWIG, G., PRETSCHER, P., SCHRÖDER, E., SSYMANK, A. (Bearb.):
Das europäische Schutzgebietssystem Natura 2000, Ökologie und Verbreitung von Arten der FFH-Richtlinie in Deutschland. Volume 1: Pflanzen und Wirbellose. -
Schriftenr. Landschaftspfl.Naturschutz, 69 (1): 426-432; Bonn - Bad Godesberg.
- DUELLI, P., WERMELINGER, B. (2005): Der Alpenbock (Rosalia alpina), Ein seltener Bockkäfer als Flaggschiff-Art, Eidg. Forschungsanstalt WSL, CH-8903 Birmensdorf, ISSN 1422-2876
- GEPP, J. (2002): Rosalia alpina L. - Österreichs Insekt des Jahres 2001, Entomol. Austriaca 5 / 2002
- HARDE, K. W. (1979) in FREUDE, H., HARDE, K. W., LOHSE, G. A.: Die Käfer Mitteleuropas, Volume 9, Phytophaga, Cerambycidae (Bockkäfer), p. 7-94, Verlag Goecke & Evers, Krefeld
- HORION, A. (1974): Faunistik der mitteleuropäischen Käfer, Volume XII
- KÖHLER, F. & B. KLAUSNITZER (1998): Verzeichnis der Käfer Deutschlands - Entomofauna Germanica, Dresden
- NIEHUIS, M. (2001): Die Bockkäfer in Rheinland-Pfalz und im Saarland, GNOR-Eigenverlag, Mainz
- SCHENKELING, S. (1917): "Erklärung der wissenschaftlichen Käfernamen aus Reitter's Fauna Germanica", K. G. Lutz' Verlag, Stuttgart
- SLÁMA, M. (1998): Tesaříkovití - Cerambycidae, Česiré republiky a Slovenské republiky (Brouci - Coleoptera), p. 95-97
- STEINER, S. (1999): Rote Liste der Bockkäfer Kärntens, Naturschutz in Kärnten 15: 269 - 286, Klagenfurt
Internet:
|





 The distribution of the Rosalia longicorn stretches over Central and Southern Europe. The western border of the range is marked by the Pyrenees. The
southwesternmost records originate from the Spanish provinces of Asturias, Cantabria, the Basque Country, Navarre, Aragon and Catalonia.
VILLERS (1946) does not mention the species for North Africa. DEMELT (1957) cites Rosalia alpina for North Africa from the Atlas Mountains,
but without mentioning a source and does also not refer to VILLERS' Fauna of North Africa - presumably out of ignorance. SAMA (2002),
a distinguished expert of the North African longhorn beetle fauna, judges the report as erroneous (BENSE, written notice). Therefore Rosalia alpina
should be deleted from the North African species list, although HORION (1974) does mention the species for North Africa.
Rosalia alpina is known to occur to the southern tip of Italy, also on the islands Sicily and Corsica, but not - contrary to other peoples' opinion - on Sardinia.
In Southeast Europe, the range stretches from Greece and Bulgaria to Southeastern Turkey, where the species is represented by the subspecies Rosalia alpina syriaca
PIC, 1894. According to SAMA (2002) the occurrences in Southeastern Turkey are probably only relics and the species might be already extinct there.
Various authors also cite Jordan, Israel, Palestine and Syria. According to the recent Fauna of Israel by SAMA (2002) and SAMA et al. (2010)
this seems to be incorrect (BENSE, written notice) and is at least questionable. In the northeast of Turkey the border of the distribution range is marked by
the Caucasus, although the beetle seems to occur around the whole Black Sea. The northern limit of the distribution runs through Northern France, where the occurrence
is only insular and isolated, through Southern Germany and Czechia to Poland. Poland also marks the northeastern limit of distribution.
In Eastern Europe, the distribution stretches over Slovakia, Hungary and the Ukraine to Russia. LAGUNOV and NOVOZHENOV reported the species in
1996 for the vicinity of Yekaterinburg (formerly Sverdlovsk) from the Ilmen natural reserve (Western Siberia, central Ural Mountains).
SHAPOVALOV collected a specimen on July 13th, 2008 in the Orenburg region in the Southern Ural Mountains (BENSE, written notice).
Hence, the Central and South Ural Mountains constitute the eastern limit of the distribution of Rosalia alpina.
According to Horion, at the beginning of the 20th century there was a settlement in Denmark on the island Sjælland and in South Sweden for a brief period of time.
The last record was made in 1926 for Västergötland.
The distribution of the Rosalia longicorn stretches over Central and Southern Europe. The western border of the range is marked by the Pyrenees. The
southwesternmost records originate from the Spanish provinces of Asturias, Cantabria, the Basque Country, Navarre, Aragon and Catalonia.
VILLERS (1946) does not mention the species for North Africa. DEMELT (1957) cites Rosalia alpina for North Africa from the Atlas Mountains,
but without mentioning a source and does also not refer to VILLERS' Fauna of North Africa - presumably out of ignorance. SAMA (2002),
a distinguished expert of the North African longhorn beetle fauna, judges the report as erroneous (BENSE, written notice). Therefore Rosalia alpina
should be deleted from the North African species list, although HORION (1974) does mention the species for North Africa.
Rosalia alpina is known to occur to the southern tip of Italy, also on the islands Sicily and Corsica, but not - contrary to other peoples' opinion - on Sardinia.
In Southeast Europe, the range stretches from Greece and Bulgaria to Southeastern Turkey, where the species is represented by the subspecies Rosalia alpina syriaca
PIC, 1894. According to SAMA (2002) the occurrences in Southeastern Turkey are probably only relics and the species might be already extinct there.
Various authors also cite Jordan, Israel, Palestine and Syria. According to the recent Fauna of Israel by SAMA (2002) and SAMA et al. (2010)
this seems to be incorrect (BENSE, written notice) and is at least questionable. In the northeast of Turkey the border of the distribution range is marked by
the Caucasus, although the beetle seems to occur around the whole Black Sea. The northern limit of the distribution runs through Northern France, where the occurrence
is only insular and isolated, through Southern Germany and Czechia to Poland. Poland also marks the northeastern limit of distribution.
In Eastern Europe, the distribution stretches over Slovakia, Hungary and the Ukraine to Russia. LAGUNOV and NOVOZHENOV reported the species in
1996 for the vicinity of Yekaterinburg (formerly Sverdlovsk) from the Ilmen natural reserve (Western Siberia, central Ural Mountains).
SHAPOVALOV collected a specimen on July 13th, 2008 in the Orenburg region in the Southern Ural Mountains (BENSE, written notice).
Hence, the Central and South Ural Mountains constitute the eastern limit of the distribution of Rosalia alpina.
According to Horion, at the beginning of the 20th century there was a settlement in Denmark on the island Sjælland and in South Sweden for a brief period of time.
The last record was made in 1926 for Västergötland.
 The male occupies a small territory on suitable breeding substrate and defends it against competing males in ritualized fights. After mating, the female deposits
its eggs singly in bark cracks and dry splittings in wood, preferably in sun-exposed areas. The total number of eggs laid by the female is unknown. The larvae
feed in the sapwood, mainly at the edge between sapwood and heartwood. They usually avoid the nutrient-poor heartwood. The development cycle takes three to four years,
but can be completed in only two years under optimal conditions. During spring and early summer the larva produces a burrow for hatching, which is subsequently
sealed with wood shavings. The arch-shaped puparium is build close under the surface and is used for pupation only. After hatching, the adult beetles reside on the breeding substrate.
They have a lifespan of three to six weeks. The beetles feed on tree sap and can also be observed feeding on foliage. On rare occasions, the beetles have been observed
on blossoms, e.g. on wild carrot (Daucus carota). Like other longhorn beetles, the Rosalia longicorn is an average flyer. On warm days the beetles swarm during the hot hours
around noon. The beetles fly around their breeding trees or search for new breeding trees or nearby logs. The spatial spreading is most probably low, in most cases the covered
distance is below 1000 m.
The male occupies a small territory on suitable breeding substrate and defends it against competing males in ritualized fights. After mating, the female deposits
its eggs singly in bark cracks and dry splittings in wood, preferably in sun-exposed areas. The total number of eggs laid by the female is unknown. The larvae
feed in the sapwood, mainly at the edge between sapwood and heartwood. They usually avoid the nutrient-poor heartwood. The development cycle takes three to four years,
but can be completed in only two years under optimal conditions. During spring and early summer the larva produces a burrow for hatching, which is subsequently
sealed with wood shavings. The arch-shaped puparium is build close under the surface and is used for pupation only. After hatching, the adult beetles reside on the breeding substrate.
They have a lifespan of three to six weeks. The beetles feed on tree sap and can also be observed feeding on foliage. On rare occasions, the beetles have been observed
on blossoms, e.g. on wild carrot (Daucus carota). Like other longhorn beetles, the Rosalia longicorn is an average flyer. On warm days the beetles swarm during the hot hours
around noon. The beetles fly around their breeding trees or search for new breeding trees or nearby logs. The spatial spreading is most probably low, in most cases the covered
distance is below 1000 m.
 In Germany, the Rosalia longicorn already disappeared in the 19th century after a short settlement from the Hannover region and Mecklenburg-Western Pomerania.
The last records from Saxony, Saxony-Anhalt and Thuringia were made before 1950. Whether these records are based on autochthonous occurrences is disputed and at least in the case
of the Hannover region and Mecklenburg-Western Pomerania doubtful (see Chapter 2 "Distribution"). From the Swabian Alb the species was recorded from the region between
Immendingen and Sigmaringen, a settlement area, from which v. d. TRAPPEN already in 1933 reported a critical threat for the species due to cutting of overaged beeches.
Today, Rosalia alpina still occurs in the above-mentioned area, but suffered severe declines since v. d. TRAPPEN until 1975.
Since then, the species at least did not disappear from the known locations in the Swabian Alb, the populations are stagnant. Since about 1993, the populations on the Swabian
Alb seem to recover. However, any spatial spreading is not discernible (BENSE et al. 2003). Records from Baden originate exclusively from the time before 1950,
although Rosalia alpina is also currently recorded from the historically known areas. The latter are assigned to Württemberg today. In Bavaria the situation looked extremely
bad until the 1980s. Until 1980, there was a loss of habitats of close to 30% observed, and another 40% during the 1980s, relative to the populated areas known in 1980.
Only around 1990 the populations became stagnant. Since around 10 years, the populations recover noticeably (BENSE et al. 2003). In the Red List of Germany
the Rosalia longicorn is classified as endangered (RL 2) (BINOT 1998).
In Germany, the Rosalia longicorn already disappeared in the 19th century after a short settlement from the Hannover region and Mecklenburg-Western Pomerania.
The last records from Saxony, Saxony-Anhalt and Thuringia were made before 1950. Whether these records are based on autochthonous occurrences is disputed and at least in the case
of the Hannover region and Mecklenburg-Western Pomerania doubtful (see Chapter 2 "Distribution"). From the Swabian Alb the species was recorded from the region between
Immendingen and Sigmaringen, a settlement area, from which v. d. TRAPPEN already in 1933 reported a critical threat for the species due to cutting of overaged beeches.
Today, Rosalia alpina still occurs in the above-mentioned area, but suffered severe declines since v. d. TRAPPEN until 1975.
Since then, the species at least did not disappear from the known locations in the Swabian Alb, the populations are stagnant. Since about 1993, the populations on the Swabian
Alb seem to recover. However, any spatial spreading is not discernible (BENSE et al. 2003). Records from Baden originate exclusively from the time before 1950,
although Rosalia alpina is also currently recorded from the historically known areas. The latter are assigned to Württemberg today. In Bavaria the situation looked extremely
bad until the 1980s. Until 1980, there was a loss of habitats of close to 30% observed, and another 40% during the 1980s, relative to the populated areas known in 1980.
Only around 1990 the populations became stagnant. Since around 10 years, the populations recover noticeably (BENSE et al. 2003). In the Red List of Germany
the Rosalia longicorn is classified as endangered (RL 2) (BINOT 1998). The Rosalia longicorn is listed on Annex II of EU Council regulation 92/43/EWG and is classified as a priority species. Hence the European Community carries a particular
responsibility for the protection and preservation of the species, also as far as its habitats are concerned. Furthermore, the species is protected by Annex II of the Berne Convention of
1979 and enjoys protection Europe-wide. That means that it is strictly forbidden to capture, kill, possess or trade beetles or parts of the beetles, as well as their development
stages or parts thereof. Also existing populations and their habitats enjoy strict protection. According to the Federal Regulation for the Protection of Species Rosalia alpina
is protected as well, as most representatives of the longhorn beetles (family Cerambycidae) are protected, except a few varmints.
The Rosalia longicorn is listed on Annex II of EU Council regulation 92/43/EWG and is classified as a priority species. Hence the European Community carries a particular
responsibility for the protection and preservation of the species, also as far as its habitats are concerned. Furthermore, the species is protected by Annex II of the Berne Convention of
1979 and enjoys protection Europe-wide. That means that it is strictly forbidden to capture, kill, possess or trade beetles or parts of the beetles, as well as their development
stages or parts thereof. Also existing populations and their habitats enjoy strict protection. According to the Federal Regulation for the Protection of Species Rosalia alpina
is protected as well, as most representatives of the longhorn beetles (family Cerambycidae) are protected, except a few varmints.
